
Orally delivered antibody drugs made commercially viable for the first time
Project Overview:
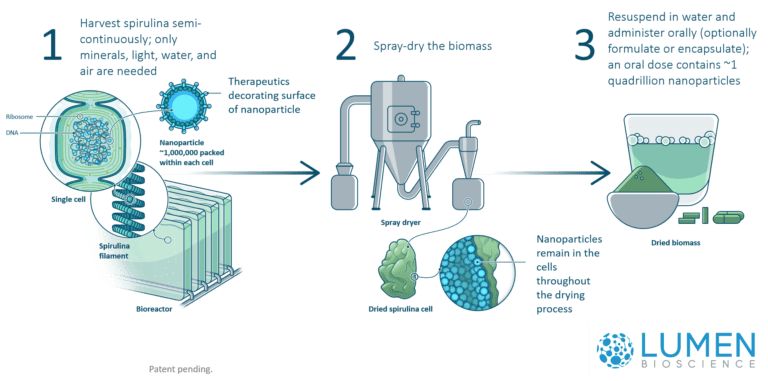
Lumen makes its biologic drugs by engineering a microbe called spirulina (Arthrospira platensis) to express the active drug molecule (most commonly an antibody) within its cell membrane. These cells are grown indoors, and the entire biomass is then dried to a powder. This powder is comprised of spirulina cells, each one filled with a therapeutic antibody "payload." This powder is then packed into dose-specific capsules, which don't require refrigeration and are shelf-stable indefinitely at room temperature.
The spirulina cells do not survive the drying process, but research shows that, when consumed, the cell membrane protects the therapeutic antibodies during transit through the acidic environment of the stomach, but conveniently releases them in the small intestine where they can bind to and neutralize the disease target.
This approach — making biologic drugs for delivery within a food substance — has been demonstrated in principle many times before but Lumen's technology makes this approach commercially viable for the first time. It was long suspected that spirulina would be a valuable tool for making biologic drugs if it could be genetically engineered. But despite decades of efforts by many well-funded groups around the world, all attempts failed. Lumen was the first to achieve this breakthrough, and as a result was recently granted a broad patent on this technology (U.S. patent #10,336,982).
_________________Platform Technology:
With this bioengineering technology in hand, the process of making a new drug is straightforward: (1) a gene encoding the therapeutic molecule (typically an antibody) is introduced into the spirulina chromosome, (2) when that strain spirulina is grown, the gene expresses the therapeutic protein inside the cell's cytoplasm, (3) the production system requires only water, mineral salts, and light so it is cheap and rapidly scalable, (4) harvesting the spirulina is done by simply spray-drying the biomass.
Lumen can manufacture orally delivered biologic drugs for 2-3 orders of magnitude lower cost than traditional biotech manufacturing systems. This is possible because:
1. being extraordinarily high in protein (>70%), spirulina cells are naturally able to express far higher amounts of therapeutic proteins than any other food crop or microbe
2. being an extremophile (pH>10), aseptic conditions are not required, and cap-ex utilization is nearly 100%
3. given spirulina's decades-long history of safe consumption, costly purification is not required — the entire biomass can be eaten whole
Importantly, Lumen has already de-risked the manufacturing elements of this project, having established a large-scale (>4,000-liter) biomanufacturing plant in late 2018. This large-scale pilot plant has been operating, under GMP conditions, for almost two years, and received third-party audit certification in early 2019.
_________________Funding Development:
All of our programs address diseases that traditional biopharma technologies have failed to solve for decades. In addition, all of them are closely associated with the increasing challenges of antibiotic resistance, and most are major causes of infant mortality and morbidity globally. Consequently, we have obtained research grants from a wide range of government agencies (e.g. NIH, USDA, Dept of Energy, US Dept of Defense) and from NGOs (e.g. Gates Foundation, Wellcome Trust).
_________________Impact:
Many chronic diseases — in both the developed and developing world — are technically solvable with modern biologic drugs, but until now the high cost of manufacturing biologics has blocked commercialization. Lumen's technology allows for the development, manufacturing, and delivery of orally delivered biologic drugs for a cost as low as small molecule drugs. This makes it feasible to treat and prevent these diseases for the first time. Following approval, Lumen has committed to the Gates Foundation that it will make its products available, at minimal markup, for distribution to infants in the developing world.
Even for retail customers from wealthier, developed-world countries, Lumen’s drastically lower cost structure enables a retail price of as little as $50 for a 2-week supply, affordable by all travelers. The enormous size of this market — there are 50-100 million travelers each year from the developed world to regions where traveler’s diarrhea is endemic — is so large that the regulatory approval costs are commercially viable, even at such a low final sales price. The impact in lives saved and economic loss avoided will be vast.
Steps | Log in -> Vote project -> Vote now -> Confirm Vote -> Be BOLD
---
VOTES

